In Tamil Nadu politics, memory is not just emotion, it is currency. And no political memory carries more weight than that of J Jayalalithaa, the late AIADMK supremo revered as “Amma,” whose political legacy continues to shape electoral calculations nearly a decade after her death.Now, former AIADMK chief VK Sasikala is attempting a political comeback through a new outfit, All India Puratchi Thalaivar Makkal Munetra Kazhagam, pitching herself as a natural inheritor of the Dravidian legacy. Her claim rests on her experience, the relationship she shared with Amma, and the argument that she alone can reclaim political space from the ruling DMK.But in the upcoming Tamil Nadu elections, will this nostalgia be sufficient to secure her votes?
The Amma legacy
Under Jayalalithaa’s leadership, governance became a carefully engineered blend of welfare delivery and emotional politics. Subsidies accounted for more than a third of Tamil Nadu’s revenue expenditure, forming the backbone of a populist model that deeply resonated with women, rural voters, and lower-income households.Her government was also marked by social interventions that strengthened her “protective leader” image. Tamil Nadu became the first state in India to allow government hospitals to perform medical procedures for transgender people to help them fight infections, a move widely seen as progressive within a traditionally conservative political environment.

From subsidised goods to targeted welfare schemes, the state’s administrative machinery often functioned as an extension of Amma’s personal brand. That direct linkage between leader and beneficiary created an unusually strong emotional voter contract, one that still influences political behaviour today.This is the legacy Sasikala is attempting to tap into, not just politically, but emotionally.
From power centre to political outsider
Sasikala’s political story is inseparable from Jayalalithaa’s inner circle. Known as “Chinnamma,” she functioned for decades as a powerful intermediary within the AIADMK ecosystem, controlling access to the leadership and influencing cadre-level decisions without holding formal office.Introduced into the AIADMK orbit in the early 1980s, she gradually became a constant presence at Jayalalithaa’s Poes Garden residence. Over time, she evolved from confidante to gatekeeper, becoming one of the most influential but unofficial figures in the party structure.After Jayalalithaa’s death in 2016, Sasikala was unanimously elevated as AIADMK general secretary during a leadership vacuum. At the time, the party was founded by MG Ramachandran, and Sasikala’s rise briefly positioned her as its central authority.Her assertion then was ambitious: “Amma is not with us now but our party will rule here for the next 100 years.”But within weeks, her political trajectory began to unravel.The years that followed saw the AIADMK transform from a vertically disciplined party into a fragmented political structure. Competing factions led by EPS and OPS attempted to assert legitimacy, eventually merging in part, but without restoring the earlier emotional unity.What was once a tightly controlled cadre machine under Jayalalithaa has now become a negotiated coalition of leaders, where authority is distributed rather than centralised.

The party that once functioned as a single-command structure under Jayalalithaa now operates through negotiated leadership balance, strong organisationally, but weaker emotionally.This fragmentation is what Sasikala is attempting to exploit, but it is also what limits her reach. The AIADMK’s “Two Leaves” symbol still commands loyalty among core voters, and organisational continuity remains intact at the booth level, even if internal cohesion is strained.In this sense, Sasikala is not entering a vacuum, she is entering a structure that has already adapted without her.
Can Amma’s emotional capital be transferred?
Sasikala’s current political strategy rests on a single assumption, that Amma’s emotional capital can be transferred to her own leadership project. But Tamil Nadu’s political history suggests otherwise.Jayalalithaa’s appeal was deeply personal and tightly centralised. Her governance created a direct leader-to-voter relationship that bypassed intermediaries. Sasikala, despite her proximity, was always an enabler, not the face of that contract.That distinction is now the core challenge of her political revival.
AIADMK’s countermove: ‘Amma oath’ politics returns
Wary of the growing “Sasikala factor,” the All India Anna Dravida Munnetra Kazhagam leadership has moved to reassert Jayalalithaa’s monopoly over political memory.Senior leader Edappadi K Palaniswami issued a call to cadres ahead of Jayalalithaa’s birth anniversary on February 24, asking them to take a pledge in her name.Cadres were instructed to light lamps at 6 pm in their homes, pray to Amma’s “atma,” and vow to protect the party till their last breath. The leadership framed it as a spiritual-political exercise, warning against “enemies and betrayers” attempting to weaken the organisation.Though Sasikala was not named explicitly, the political signal was unmistakable — a pre-emptive mobilisation against internal and external challengers.The directive came amid heightened political churn triggered by Sasikala’s re-entry into Tamil Nadu politics after her prison release, and renewed activity by her nephew TTV Dhinakaran, both of whom the AIADMK expelled in 2017.
DMK’s consolidation: Governance over nostalgia
At the same time, the ruling Dravida Munnetra Kazhagam under Chief Minister MK Stalin has consolidated its position through welfare expansion, institutional governance, and cadre stability.Unlike personality-driven mobilisation, the DMK’s strategy increasingly relies on scheme-based outreach and bureaucratic delivery systems that reduce dependence on emotional recall.Moreover, the entry of O Panneerselvam into the DMK camp led by MK Stalin marks a significant political shift, especially in the battle over the legacy of J Jayalalithaa. Once a loyalist and symbolic inheritor of her political legacy within the AIADMK, OPS is now positioning himself alongside Stalin, even invoking the blessings of both Amma and MG Ramachandran to back the DMK’s return to power. This move not only sharpens his rivalry with Sasikala. In doing so, OPS is recasting himself from an internal dissenter to a broader political player, leveraging the emotional weight of the Amma legacy in a new alliance ahead of the 2026 elections.
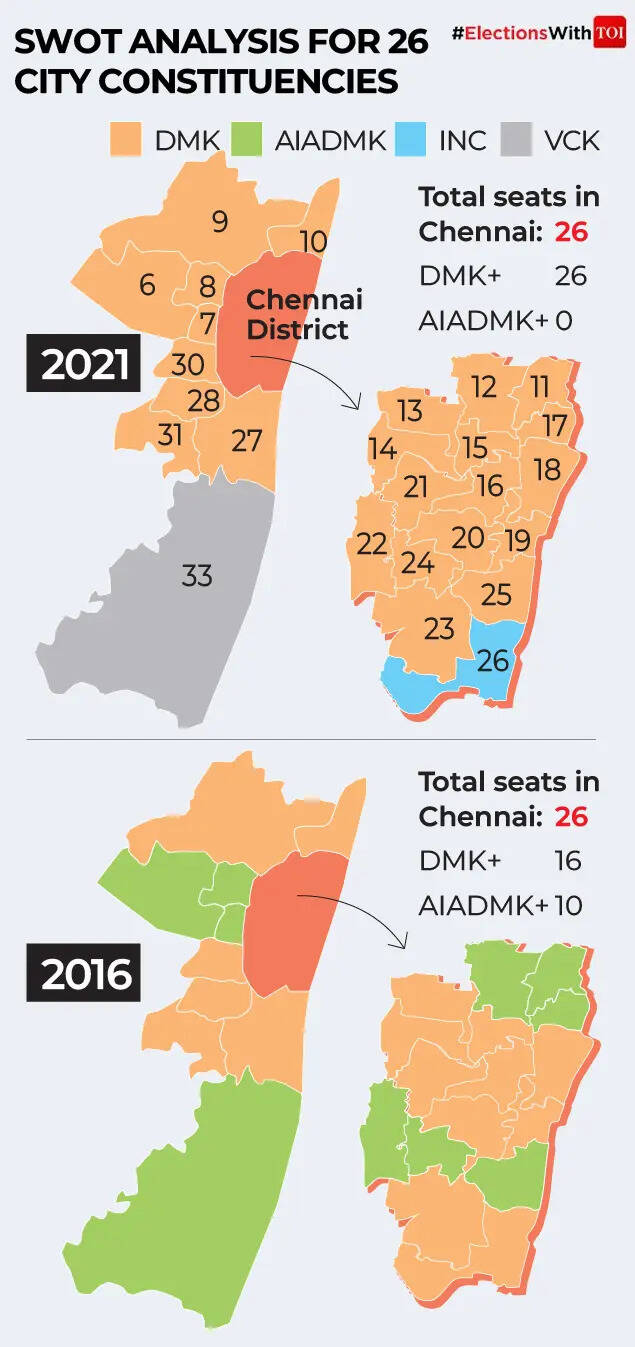
A crowded opposition: Vijay factor and vote splitting
The entry of actor-politician Vijay through Tamilaga Vettri Kazhagam has further reshaped the battlefield.What was once a bipolar contest is now a multi-polar field, where anti-incumbency sentiment is dispersed across multiple challengers. This fragmentation reduces the probability of any single opposition force converting emotional appeal into electoral dominance.In this environment, even strong emotional narratives risk being diluted into overlapping voter bases rather than consolidated support blocs.On the campaign trail, Sasikala has leaned heavily on Jayalalithaa-era symbolism, invoking Amma, referencing MGR, and projecting herself as part of the original Dravidian ideological lineage.Her party flag features Dravidian icons, reinforcing continuity with political tradition rather than rupture. But political observers note a critical gap: symbolism does not automatically translate into cadre mobilisation.Jayalalithaa’s rallies were defined by precision, command, and tightly controlled messaging that turned emotion into electoral machinery. Sasikala’s current political style, though emotionally charged, lacks the same disciplined mass orchestration.Sasikala’s return is also framed by ongoing legal and political aftershocks. Civil suits filed by her and Dhinakaran challenging AIADMK organisational decisions remain pending. Additionally, state-level actions related to disproportionate assets investigations, including confiscation of properties linked to her relatives, continue to cast a shadow over her political rehabilitation.These unresolved disputes ensure that her political comeback is being fought simultaneously in courtrooms, party structures, and public perception battles.There is no doubt that Amma nostalgia continues to influence Tamil Nadu’s political psyche. It is visible in welfare memory, symbolic politics, and emotional recall across sections of the electorate.But nostalgia is not a standalone vote bank. It requires organisational depth, leadership credibility, and electoral machinery to convert sentiment into seats.For Sasikala, the challenge is not simply invoking Amma’s legacy, it is proving she can operate outside it. The AIADMK is invoking Amma to protect itself. The DMK is countering it through governance. And Sasikala is attempting to claim it as an inheritance.

In between lies the central question of Tamil Nadu politics today – can memory survive without machinery, and can machinery be rebuilt from memory alone?